 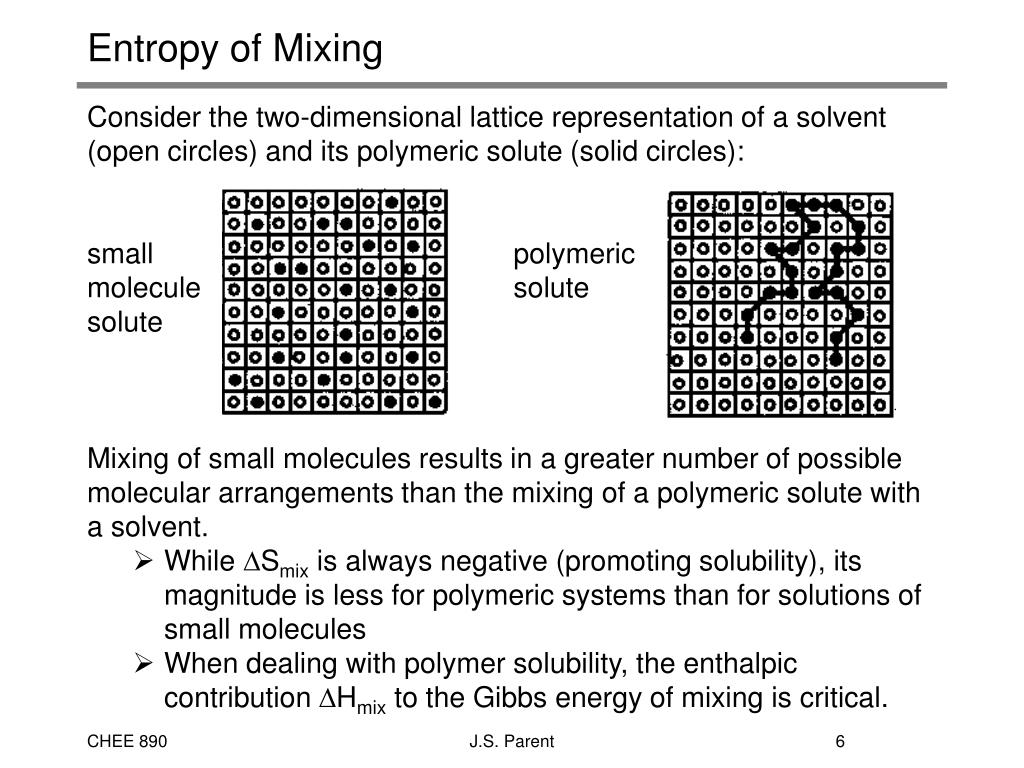
Vinegar, an aqueous solution (mostly water), is considered a "polar" material whose molecules have a relatively large electric dipole moment. What are we missing? Considering orientation But the second law of thermodynamics predicts an increase in entropy. Obviously, the entropy doesn't decrease during phase separation. The analysis so far shows no change in entropy for mixed and phase separated vinaigrette. Since log 1 = 0, there is no entropy in either volume. There is only one way to arrange indistinguishable particles into either the mixed or phase-separated state. In both macrostates, oil and vinegar are found in positions of zero uncertainty. The phase-separated equilibrium condition and a lightly mixed dressing (approaching equilibrium, with just a single drop of oil phase-separated from the bulk) are shown on the left and right, respectively. Vinaigrette dressing (a liquid) is densely packed with molecules with no free volume - each square must contain either an oil or vinegar particle. Both systems are filled with a standard recipe of 2/3 oil and 1/3 vinegar by volume. Specifying macrostatesĪs an example, consider a grid with 12 rows and 12 columns (144 total squares) with oil and vinegar particles represented in black and white, respectively. Since the volumes are discrete, the number of microstates is countable. We can understand the distinction between macroscopic and microscopic conditions using a simple 2D lattice in which a macroscopic system is modeled as a rectangular grid filled with squares, each representing a microscopic volume. It may seem ironic, but it’s a conclusion based on the fundamentals of statistical physics. Somehow, the mixed condition has lower entropy than the stable, fully phase separated condition. The second law of thermodynamics tells us that the entropy of isolated, spontaneously changing systems, including vinaigrette, must increase. It's easy to conflate the macroscopic randomness of mixed conditions with disorder at the microscopic level.Įxperience tells us that vinaigrette will spontaneously separate into oil and vinegar if left undisturbed. Correspondingly, mixed dressing can appear in many different, seemingly random configurations. This unique condition appears highly ordered on the macroscopic scale. Higher entropy macroscopic conditions are more disordered only on the microscopic scale.Ĭonsider the vinaigrette salad dressing at thermal equilibrium with fully phase separated liquids. According to Boltzmann, entropy is defined as a measure of (proportional to the logarithm of) the number of microstates that are consistent with the macroscopic conditions of the system. These include volume, mass, density, energy, momentum, temperature and other properties associated with large collections of molecules. What we can observe are macroscopic conditions. Microstates are essentially a figment of our imagination. Simply stated, we can’t observe microstates. Measuring the position and momentum of every individual molecule in a group would also change the state into some other, unknown condition (see Heisenburg Uncertainty Principle). Even with the highest resolution instruments, we can't directly observe microstates. Ludwig Boltzmann defined entropy in terms of "microstates" that describe the position and momentum of every particle in the system. Why is this confusing? Microscopic disorderĮntropy is a measure of microscopic disorder (on the sub-molecular-length scale). While this may seem plausible (the oil and vinegar solutions appear increasingly more organized) a decrease in entropy would violate the second law of thermodynamics. One misconception is that the entropy of the mixture decreases with time as the liquids separate into pure phases. Stable, fully phase-separated dressing has higher entropy than mixed. This is the case with vinaigrette, where the entropy of the dressing increases as it separates. Entropy and the second law of thermodynamicsĪccording to the second law of thermodynamics, the entropy of an isolated system undergoing spontaneous transition increases until it reaches a maximum value at thermal equilibrium. If left isolated and undisturbed, vinaigrette mixtures can spontaneously separate into stratified layers of oil on water. The two liquids are immiscible (difficult to mix), and form distinct, separate volumes of oil and vinegar phases. Liquid-liquid phase separation is a phenomenon commonly observed in vinaigrette salad dressing, which is made from a mixture of oil and vinegar. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
